Background
Worldwide, more than 1 400 000 men were diagnosed with prostate cancer in 2020, and it is estimated that one third will need radiotherapy during their treatment.
Following Standards
Accuracy and Precision are required for quality treatment. Accuracy refers to the closeness to the true or accepted value and precision refers to the closeness between repeated measurements. There are many factors that can influence the quality of the radiotherapy, but the delineations of the target volumes and the organs at risk are essential. Accuracy of delineation is related to following the generally accepted standard (the contouring guidelines) and precision is provided when the differences in the contouring style are minimized.
Inter-Observer Variations
When 120 contours made by 6 radiation oncologists were compared to a “gold standard” (defined using data from the Visible Human Project), systematic delineation errors were observed – the posterior portions of the prostate were always missed, while anteriorly, some normal tissue was always defined as target.
A recently published paper analyzed the contours of 492 prostate cancer treatment plans created by 4 Canadian radiation oncologists working in the same department. Significant variability in the contouring of target volumes was detected for both prostate and pelvic lymph nodes CTV structures. In the high-risk patients’ cohort, significant variations were observed for rectum length and penile bulb volumes and lengths.
Contouring Variation Impact on the DVH
In the Canadian study, there were significant variations in mean DVHs of the organs at risk.
A multi-institutional contouring challenge in which 25 observers delineated prostate, rectum, and bladder on the same CT scan analyzed the corresponding DVHs, demonstrating that the variance in prostate DVH is primarily driven by differences in prostate contouring. Observed rectal DVH variation was primarily driven by differences in rectal contouring as well as prostate contouring.
Delineation Accuracy, Precision, and Speed
Automated segmentation looks tempting. It definitely works faster, but is the result good enough? Does it require extensive editing?
We can choose the atlas-based auto-segmentation or the Artificial Intelligence-driven models.
When compared, multiple-atlas-based strategies have better accuracy than single-atlas approach. However, when comparing atlas-based with AI-based auto-contour, the accuracy was higher for the AI-based models, and also the time necessary for editing the result was significantly lower.
The first version of the MVision prostate model was created based on 900 CT scans. It was evaluated by clinicians in a multicentric, retrospective study. Compared to manual contouring, the mean time saved using the MVision automated segmentation tool was 12 minutes for the whole data set (46%) and 12 min for the lymph node CTV (60%), respectively. The inter-observer variation was evaluated for the manual contours and then for the edited automatic contours. The delineation consistency, DICE similarity coefficient (DSC) improved from 0.78 to 0.94 for the whole data set and from 0.76 to 0.91 for the lymph nodes.
You can find out more and you can test it, too!
MVision’s guideline-based segmentation solution, GBS™, improves standardisation and increases consistency. Our GBS™Male Pelvis model features 34 ROIs and 2 lymph node levels, following consensus guidelines including RTOG and PIVOTAL.
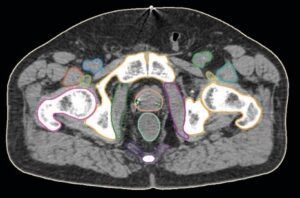
Here is a preview of our prostate model.
MVision’s Guideline-Based SaaS Solution
MVision AI is the pioneer of guideline-based AI segmentation, GBAIS™, radiotherapy software. Our deep learning system helps you automate contouring, including lymph node levels, to streamline your cancer treatment planning. Our software complies with HIPAA, and GDPR, bringing the highest quality radiotherapy planning SaaS solution for your oncology department.
Book a demo to explore guideline-based segmentation for your clinic.
MVision AI is active on LinkedIn, Twitter, Facebook, and Instagram. Follow us on social media and subscribe to our newsletter to stay up-to-date on the latest MVision AI news.
Are you eager to learn more? We welcome you to contact MVision AI:
c/o Terkko Health Hub, Haartmaninkatu 4, 00290 Helsinki, Finland.
Tel: +358 (0) 40 5489 229
Email: info@mvision.ai
For media inquiries:
+358 40 500 7915; pr@mvision.ai
Sources
Wang, L., Lu, B., He, M., Wang, Y., Wang, Z., & Du, L. (1AD, January 1). Prostate cancer incidence and mortality: Global status and temporal trends in 89 countries from 2000 to 2019. Frontiers. Retrieved June 17, 2022, from https://www.frontiersin.org/articles/10.3389/fpubh.2022.811044/full
Prostate cancer treatment statistics. Cancer Research UK. (2021, February 5). Retrieved June 17, 2022, from https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/prostate-cancer/diagnosis-and-treatment
Accuracy and precision. (2022, May 23). Retrieved June 17, 2022, from https://en.wikipedia.org/wiki/Accuracy_and_precision
Song, S., Guo, Y., Yang, Y., & Fu, D. (2022, March 10). Advances in pathogenesis and therapeutic strategies for osteoporosis. Pharmacology & Therapeutics. Retrieved June 17, 2022, from https://www.sciencedirect.com/science/article/abs/pii/S0163725822000626
Patrick, H. M. (n.d.). Reduction of inter-observer contouring variability in daily clinical practice through a retrospective, evidence-based intervention. Taylor & Francis. Retrieved June 17, 2022, from https://www.tandfonline.com/doi/full/10.1080/0284186X.2020.1825801
Barghi, A., Johnson, C., Warner, A., Bauman, G., Battista, J., & Rodrigues, G. (2013, November 5). Impact of contouring variability on dose-volume metrics used in treatment plan optimization of PROSTATE IMRT. Cureus. Retrieved June 17, 2022, from https://www.cureus.com/articles/2348-impact-of-contouring-variability-on-dose-volume-metrics-used-in-treatment-plan-optimization-of-prostate-imrt
DL;, W. W. K. L. L. H. K. (n.d.). Evaluation and optimization of the parameters used in multiple-atlas-based segmentation of prostate cancers in radiation therapy. The British journal of radiology. Retrieved June 17, 2022, from https://pubmed.ncbi.nlm.nih.gov/26539630/
Comparative clinical evaluation of auto segmentation methods in contouring of prostate cancer. (n.d.). Retrieved June 17, 2022, from https://www.oncologyradiotherapy.com/articles/comparative-clinical-evaluation-of-auto-segmentation-methods-in-contouring-of-prostate-cancer.pdf
Clinical evaluation of deep learning and Atlas. (n.d.). Retrieved June 17, 2022, from https://www.practicalradonc.org/article/S1879-8500(20)30161-2/fulltext