Radiotherapy is an important modality used for the treatment of lung cancer. Nearly 80% of all patients with lung cancer have an evidence-based indication for radiotherapy. (Vinod, Shalini K., and Eric Hau.) If evidence-based radiotherapy recommendations were followed by the entire population of lung cancer patients, it is estimated that radiotherapy alone would result in a 4% increase in overall patient survival. (Shafiq, J., et al.) This article will discuss new research in cancer care and radiotherapy treatment both overall and as an effective method to treat lung cancer. Furthermore, this article will examine the use of stereotactic body radiation therapy (SBRT) as an innovative treatment for lung cancers.
2021 ASTRO-presented research on radiotherapy and lung cancer treatment overall
Using computer modeling to define and predict individual tumor responses for improved radiocurability
Radiation treatment is one of the most frequently used therapies against cancer. However, advances in radiation oncology have primarily focused on improving physical dose properties, resulting in a lack of individualized radiation dose prescription based on the biology of a patient’s tumor. (Alfonso, Juan C. L., et al.) Researchers at Moffitt Cancer Center working to improve the personalization of radiation therapy through computer modeling.
The researchers developed a computer model to study the relationship between the tumor-immune environment and a tumor’s response to radiotherapy. The model is a silico 3-dimensional agent-based model of diverse tumor-immune ecosystems (TIES) represented as anti- or pro-tumor immune phenotypes. (Alfonso, Juan C. L., et al.) The model included factors the following factors:
- Cell proliferation
- Migration
- Cell death
- Immune cell motility
- Cell to cell interactions
- The cytotoxic impact of radiation
From the model, the research team discovered that tumor cells will either escape immune predation or be eradicated by the immune system based on the numbers of immune effector and suppressor cells present.
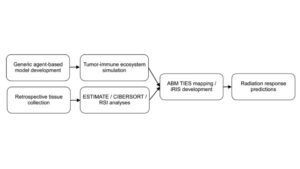
The research team analyzed samples from 10,469 patients across 31 tumor types and were able to estimate tumor, immune effector, and immune suppressor cell numbers in each tumor. The results were mapped into the model, displaying the potential for radiotherapy to alter the immune environment for maintaining tumor eradication, while the probability for other tumors that did not experience this alternative to regrow is likely. (Alfonso, Juan C. L., et al.)
“The team at Moffitt combined the factors that predicted these radiation-induced shifts into an individual Radiation Immune Score (iRIS) and used the model to analyze data from non-small cell lung cancer (NSCLC) patients who received radiation and demonstrated that the iRIS score correlates with tumor responses to radiation and the overall survival of the patients. The researchers found that the combination of the iRIS values with a second biomarker that estimates tumor radiosensitivity called the radiosensitivity index (RSI) stratifies patient responses to radiation therapy, with low RSI and low iRIS scores being associated with enhanced radiocurability.” (Moffitt Cancer Center)
Implementing ACCURE Intervention to prevent racial disparities for cancer patients
African American patients with curable lung or breast cancer complete treatment less often than similar caucasian patients. This, in turn, contributes to a worse survival rate for African American cancer patients. (Manning, M., et al.) There was evidence to suggest that ACCURE, a system-based intervention trial, helped to eliminate black-white disparities in cancer treatment completion.
The ACCURE intervention consists of multiple parts:
- An electronic health record with automatic alerts to flag missed appointments or unmet milestones in expected care (The ASCO Post)
- A nurse navigator trained in race-specific barriers to help patients overcome obstacles to care when alerts are flagged (The ASCO Post)
- A physician champion to engage healthcare teams with race-related feedback on treatment completion (The ASCO Post)
- Regular health equity education training sessions for staff. (The ASCO Post)
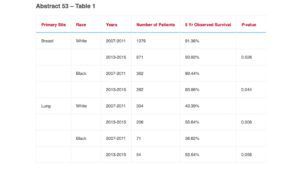
A recent, five-year trial was presented at the American Society for Radiation Oncology (ASTRO) Annual Meeting in October 2021, investigating how the ACCURE intervention influenced survival disparity. The trial included nearly 1,500 patients with stage 0, I & II lung and breast cancer diagnosed in 2013-2015, and compared their results to over 2,00 patients treated in 2007-2011. The research found statistically significant improvement for the survival for black and white breast and white lung cancer patients over time, while the racial gap in survival was reduced for lung and breast cancer.
The 5-year observed survival rate for both African American and Caucasian lung cancer patients increased on average by roughly 15%. Furthermore, subgroup analysis of lung cancer patients showed a 5-year survival rate for patients treated with SBRT by up to 50% for African American and Caucasian patients. This study suggests that equity in treatment may be associated with the elimination of survival disparities. (Manning, M., et al.)
Among the findings, half of the patients were predicted to have durable tumor control with a lower radiation dose, whereas 40% needed a higher radiation dose. The researchers suggest that the model has the potential to simulate how individual patients would respond to radiation and predict the minimal radiation dose required to achieve tumor control with and without help from immune targeting agents. (Moffitt Cancer Center)
SBRT for lung cancer treatment
The safety and effectiveness of SBRT for oligometastases
Stereotactic body radiation therapy (SBRT), also known as stereotactic ablative radiation therapy (SABR), is a cancer treatment that delivers extremely precise and intense doses of radiation to cancer cells while minimizing damage to healthy tissue. This treatment is becoming a rapidly accepted practice for radiotherapy of certain tumors.
Oligometastases refer to a state of disease where cancer has spread beyond the primary site but is not yet widely metastatic. (Hellman, S., and R. R. Weichselbaum.) A recent non-randomized phase II trial, conducted by the University of British Columbia, was presented. During the trial, all participants received experimental SABR treatment to all sites of newly diagnosed or progressing oligometastatic disease. They assessed 200 patients’ toxicity associated with this experimental treatment. Among the results, the study found a 95% confidence in the risk of late grade 4 toxicity, anticipating a < 5% rate of grade 4 toxicity. (Olson, Robert, et al.)
“It looks like toxicity from SABR provided in this provincially coordinated, and peer-reviewed setting, appears to be safe with a less than 5% grade 3 to 5 toxicity rates,” said Robert Olson, associate head of research in the department of surgery, and the head of the Division of Radiation Oncology at the University of British Columbia
The long-term benefit of SABR for operable, early-stage NSCLC
A new study was conducted by the research team at the University of Texas MD Anderson Cancer Center to investigate the long-term benefits of SBRT for early-stage, non-small-cell lung cancer (NSCLC). The findings from the single-arm, non-randomized STARS trial.
The study was formulated on the pooled analyses of two randomized studies, published in 2015, which investigated the advantages of SABR versus a surgical procedure called video-assisted thoracoscopic surgical lobectomy with mediastinal lymph node dissection (VATS L-MLND). Participants of the latest study included 80 newly diagnosed patients with early-stage NSCLC with tumor sizes 3 cm or smaller. The participants’ results were compared to propensity-matched patients undergoing surgery during the same period. A final follow-up occurred in 2020. (Chang, Joe Y., et al.)
“The results showed that the overall survival (OS) rates of the SABR and surgical cohorts were not significantly different. The SABR and surgical cohorts both achieved a three-year OS rate of 91%. The five-year OS rate was 87% in the SABR arm versus 84% in the surgical arm. The progression-free survival (PFS) rates also were similar. The three-year PFS rate was 80% for the SABR group versus 88% for the surgical group, while the five-year PFS rate was 77% versus 80%, respectively. SABR was well-tolerated, with no grade 4-5 toxicity. Only one case (1.3%) of grade 3 dyspnea was reported.” (Chien, Shanley.)
Overall, these findings illustrated that stereotactic ablative radiotherapy (SABR) was as effective as surgery at providing long-term benefits to patients with operable early-stage non-small cell lung cancer (NSCLC) and generated minimal side effects. (Chang, Joe Y., et al.)
“While surgical resection provides additional information about mediastinal lymph node involvement, the procedure is associated with significant side effects and complications,” said Joe Chang, M.D., Ph.D., professor of Radiation Oncology at the University of Texas MD Anderson Cancer Center. “Considering much lower treatment-related side effects and complications as compared with surgery, non-invasive SABR can offer patients a much better quality of life, particularly for senior patients or those with comorbidities.”
Guideline-based AI segmentation, GBAIS™, software for your clinic
MVision AI is the pioneer of guideline-based AI segmentation, GBAIS™, radiotherapy software. Within 3 months of the NHS England’s commissioning of SBRT treatments, MVision developed a lung CT, auto-segmentation model which follows the SABR Consortium guideline.
We look forward to using our deep learning system to help you automate organ-at-risk contouring, including lymph nodes, to streamline your cancer treatment planning, as we are pending 510(k) FDA clearance. Robust HIPAA, and GDPR compliant, we bring the highest quality radiotherapy planning SaaS solution for your oncology department.
MVision AI is active on LinkedIn, Twitter, Facebook, and Instagram. Follow us on social media to stay up-to-date on the latest MVision AI news.
Are you eager to learn more? We welcome you to contact MVision AI:
c/o Terkko Health Hub, Haartmaninkatu 4, 00290 Helsinki, Finland.
Tel: +358 (0) 40 5489 229
Email: info (at) mvision.ai
For media inquiries:
+358 40 500 7915; pr (at) mvision.ai
Sources
Alfonso, Juan C. L., et al. “Tumor-Immune Ecosystem Dynamics Define an Individual Radiation Immune Score to Predict Pan-Cancer Radiocurability.” Neoplasia, vol. 23, no. 11, 2021, pp. 1110–22. Crossref, doi:10.1016/j.neo.2021.09.003.
Barton, Michael B., et al. “Estimating the Demand for Radiotherapy from the Evidence: A Review of Changes from 2003 to 2012.” Radiotherapy and Oncology, vol. 112, no. 1, 2014, pp. 140–44. Crossref, doi:10.1016/j.radonc.2014.03.024.
Chang, Joe Y., et al. “Stereotactic Ablative Radiotherapy for Operable Stage I Non-Small-Cell Lung Cancer (Revised STARS): Long-Term Results of a Single-Arm, Prospective Trial with Prespecified Comparison to Surgery.” The Lancet Oncology, vol. 22, no. 10, 2021, pp. 1448–57. Crossref, doi:10.1016/s1470-2045(21)00401-0.
Chien, Shanely. “Long-Term Benefit of SABR for Operable Early-Stage NSCLC Shown in New Study.” ScienceDaily, Science Daily, 2021, www.sciencedaily.com/releases/2021/09/210914100059.htm#:%7E:text=The%20results%20showed%20that%20the,84%25%20in%20the%20surgical%20arm.
Hellman, S., and R. R. Weichselbaum. “Oligometastases.” Journal of Clinical Oncology, vol. 13, no. 1, 1995, pp. 8–10. Crossref, doi:10.1200/jco.1995.13.1.8.
Manning, M., et al. “Overall Survival From a Prospective Multi-Institutional Trial to Resolve Black-White Disparities in the Treatment of Early Stage Breast and Lung Cancer.” International Journal of Radiation Oncology*Biology*Physics, vol. 111, no. 3, 2021, p. S28. Crossref, doi:10.1016/j.ijrobp.2021.07.091.
Moffitt Cancer Center. “Moffitt Researchers Use Computer Modeling to Predict Patient Tumor Responses to Radiation Therapy.” Moffitt Cancer Center, 2021, moffitt.org/newsroom/press-release-archive/moffitt-researchers-use-computer-modeling-to-predict-patient-tumor-responses-to-radiation-therapy.
Olson, Robert, et al. “Population-Based Phase II Trial of Stereotactic Ablative Radiotherapy (SABR) for up to 5 Oligometastases: SABR-5.” BMC Cancer, vol. 18, no. 1, 2018. Crossref, doi:10.1186/s12885-018-4859-7.
The ASCO Post. “ACCURE Trial Aims to Eliminate Disparities in Survival in Black and White Patients With Early-Stage Lung and Breast Cancers – The ASCO Post.” The ASCO Post, 2021, ascopost.com/news/october-2021/accure-trial-aims-to-eliminate-disparities-in-survival-in-black-and-white-patients-with-early-stage-lung-and-breast-cancers.
Vinod, Shalini K., and Eric Hau. “Radiotherapy Treatment for Lung Cancer: Current Status and Future Directions.” Respirology, vol. 25, no. S2, 2020, pp. 61–71. Crossref, doi:10.1111/resp.13870.


